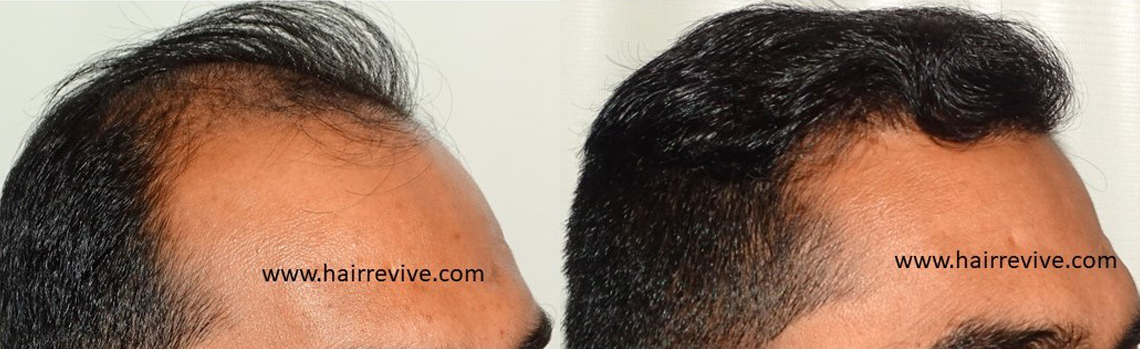
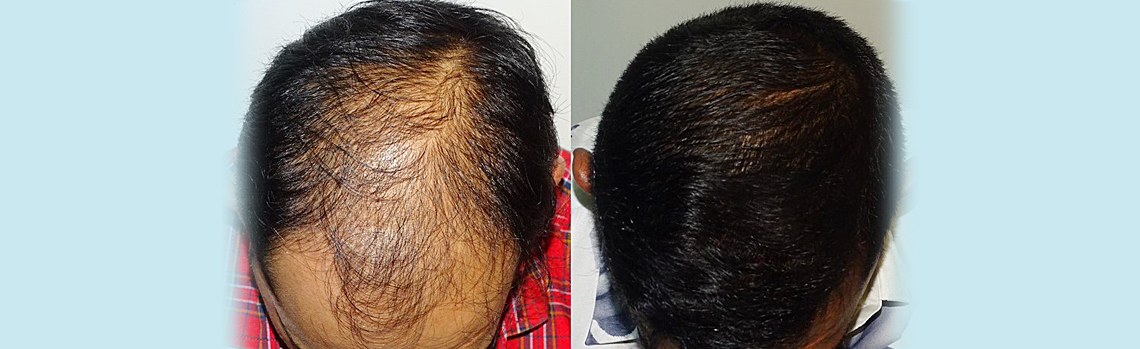
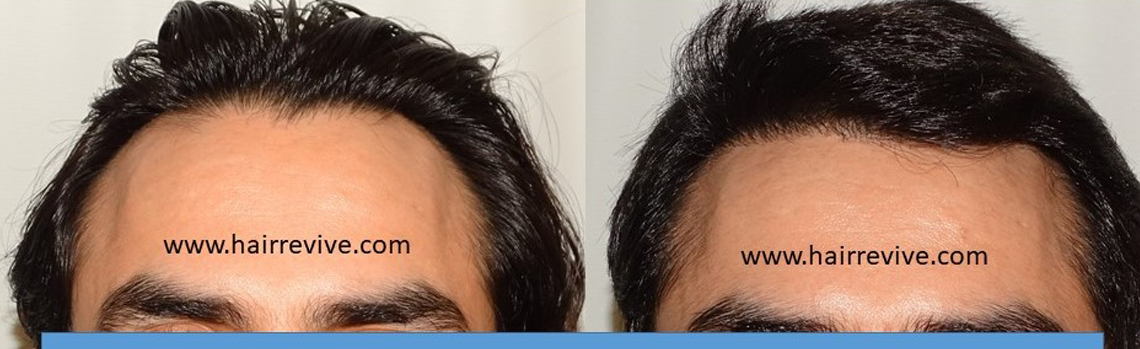
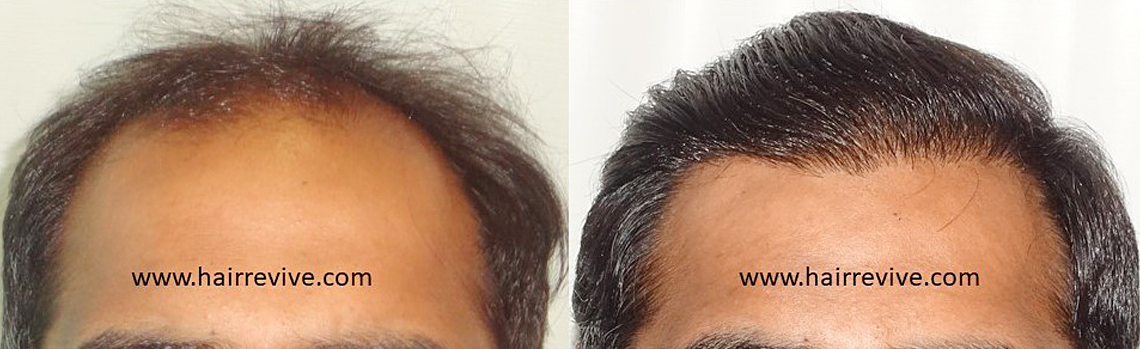
Consultation for Hair – Register for ONLINE/REMOTE CONSULTATION
Consultation for Skin Aesthetics – Register for ONLINE / REMOTE CONSULTATION
In-Person Consultation – Register here
Welcome to Hairrevive – Centre for hair restoration and Skin rejuvenation where qualified doctors provide various services including consultations and hair transplant surgeries. Dr Indu Sattur (Medical Director, Trichologist, Hair Transplant Surgeon & Cosmetologist) and Dr Sandeep Sattur (Founder, Hair Transplant & Plastic Surgeon) are heading this centre.
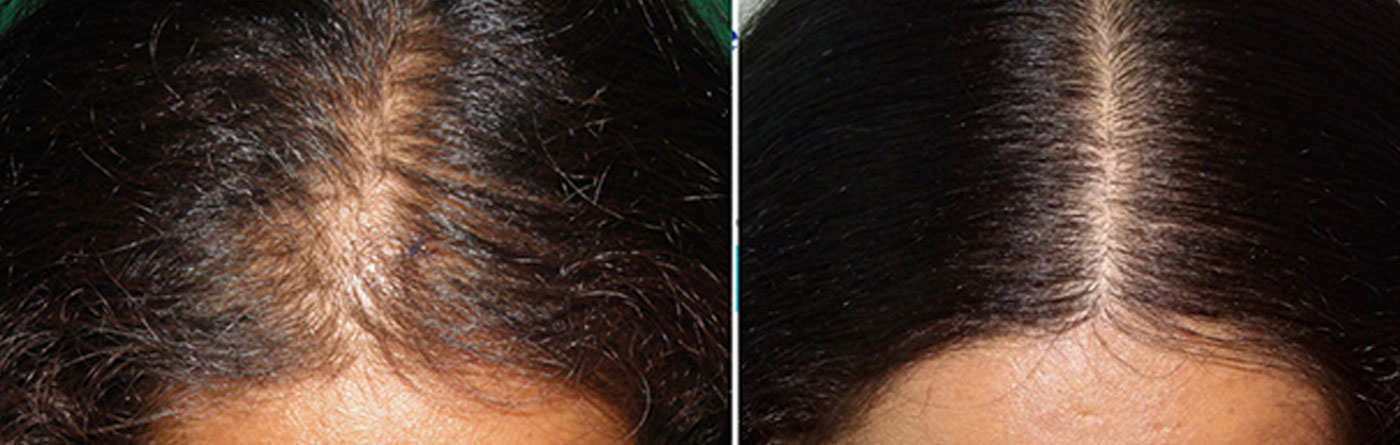
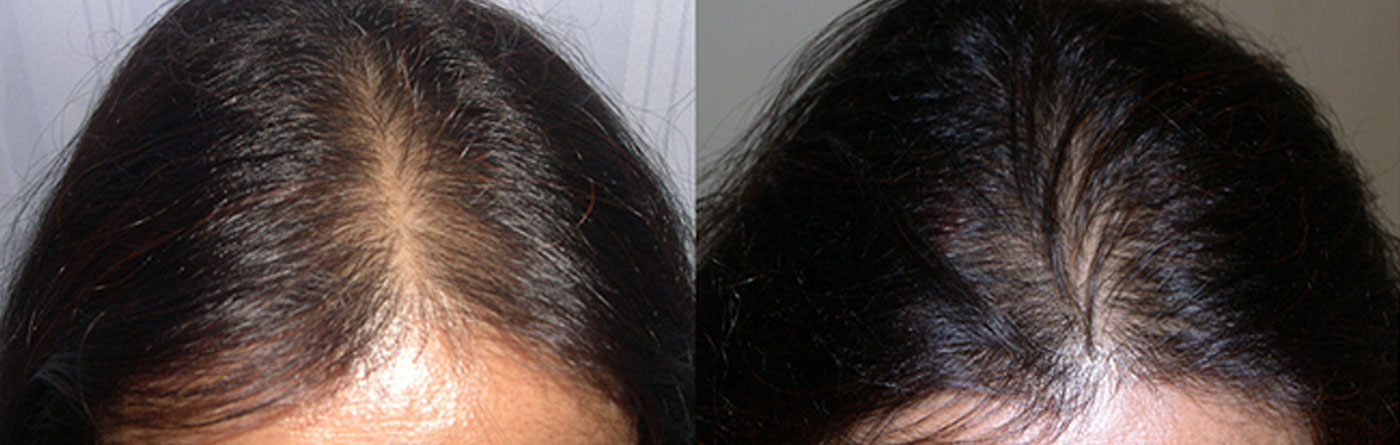
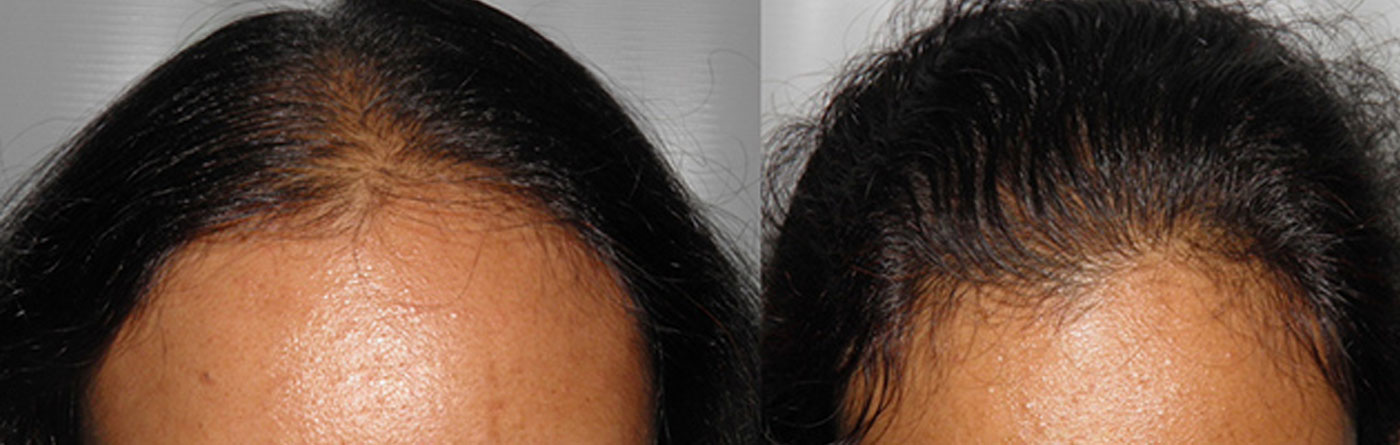
Standard of care at Hairrevive.
All consultations are done by Dr Indu and Dr. Sandeep Sattur. There are no executives/managers who consult patients. During the consultation a detailed history is taken and a thorough evaluation of the scalp and hair is made. The changes in hair shaft diameter and density seen with hair loss is evaluated with trichoscopy. If necessary additional blood tests and scalp biopsy are recommended.
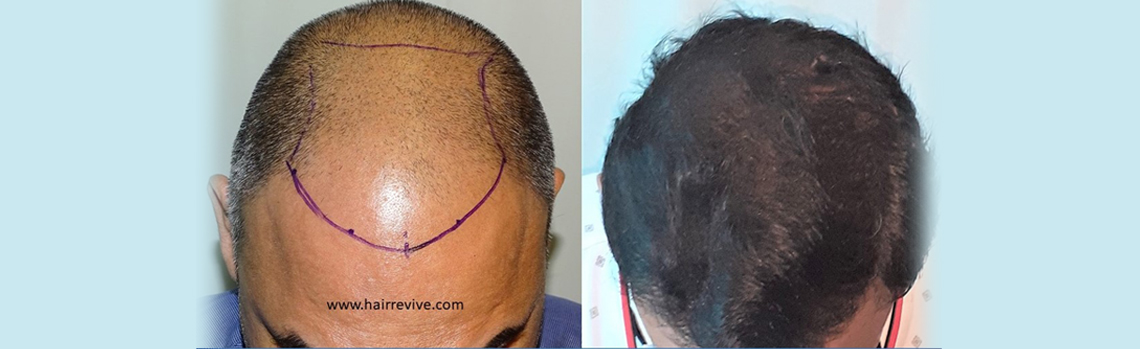
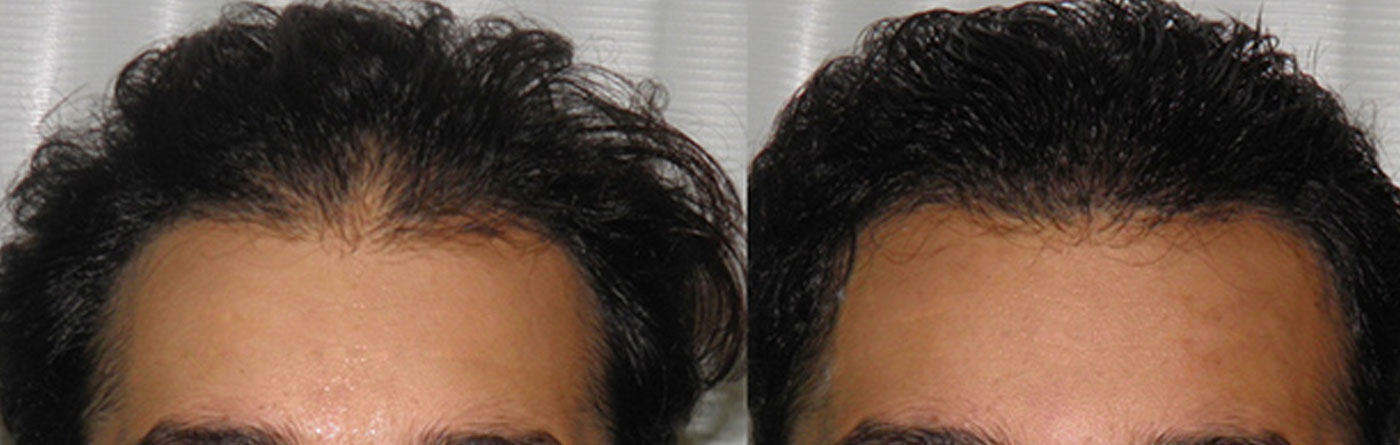
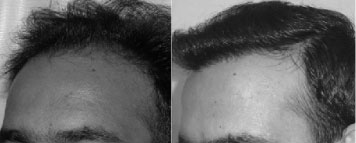
Hair transplantation
This is one of the few centres in the world where the hair transplant surgeon’s involvement is consistent in all steps of the process.
Dr. Sandeep Sattur along with Dr. Indu Sattur are responsible for the all steps leading up to the hair transplantation including detailed planning, hair line placement (in conjunction with the patient’s expectations), trimming of the hair.
During the procedure, the delivery of anesthesia and donor harvest is entirely performed by DRs Sattur and not delegated to technicians or assistants. This is one of the few centres in the world where most of the implantation is performed by the hair transplant surgeon and not technicians. Part of the implantation performed by technician is in the recipient sites created by Dr Sattur and is completely supervised by Dr Sattur. Dr Sattur is with the patient throughout the surgery. Only those tasks that can be delegated to trained technicians like separating the grafts, assisting the surgeons during extraction and implanting in recipient sites made by Dr Indu or Dr Sandeep Sattur is allowed.
Patient safety
Hair transplant surgery is an extremely safe skin surgery and the actual risk of emergency events occurring during the procedure is rare. But as the surgery is performed in an office setting it is important for the hair transplant surgeon and his team to be prepared for an emergency. This preparation is at two levels – training and infrastructure level. Training in emergency response is important for the hair transplant surgeon as in most situations he would be only person capable of performing this task during a hair transplant procedure. The American Heart Association has developed basic life support (BLS) and advanced life support (ACLS) protocols which are well thought out, systematic approaches to management of any deteriorating patient. They focus on detecting and treating life-threatening respiratory and circulatory compromise. Dr Indu Sattur and Dr Sandeep Sattur have both completed the BLS and ACLS courses.
Before the patient is taken up for surgery, a detailed assessment with blood tests and ECG (additional tests would be advised if the patient has any medical co-morbidities) is performed to rule out the patient’s fitness to undergo hair transplantation under local anesthesia. Additionally, the patient is referred to a Physician for opinion regarding patient’s fitness to undergo hair
transplantation.
A qualified anaesthetist is always present from start till the very end of the hair transplant procedure to ensure complete safety for the patient. More over during the procedure continuous monitoring is performed of the patient’s vitals. Regular record of the amount of anesthesia used is maintained.
At an infrastructure level right from the outset when this centre was started all equipment for emergency resuscitative (including a defibrillator) management and monitoring have been in place.
Emergency drugs needed in a situation are always handy. In the rare event of unexpected serious or potentially serious developments during or immediately after a hair transplant the transfer of the patient to a nearby hospital could be arranged.
Hair Restoration experience at HAIRREVIVE
-
Consultations done by DR. Sandeep Sattur and DR. Indu Sattur only
-
Preparatory steps of the hair transplant surgery including planning and marking, trimming performed by DRs Sattur
-
Anesthesia, donor harvesting and implantation done by DRs Sattur
-
Presence of a qualified anaesthetist throughout the procedure
-
Presence of all necessary monitoring and resuscitative equipment in case of any emergency present in the operation theatre
-
Follow-up assessments done by Drs Sattur
LIVE With Trichologist Dr. Sandeep Sattur
Case Studies / Stories
Mr. PD (Chicago)
“A LINER ABOUT THE CASE” Hi Dr. Sattur. It’s been a week since my hair procedure with you in Mumbai. The recovery has been going well and I am back to the grind of my every day routine. I want to take this opportunity to thank you and your dear wife who went the extra mile to accommodate me given my short stay in Mumbai. Your team was just the best. Their welcoming smiles and services made me feel right at home. Your Anesthesiologist was very efficient and friendly and kept me comfortable thought out the procedure. Please convey my thanks to her. Your honesty throughout my consultations was what impressed me the most and made me be more realistic about my expectations.
Hi Dr. Sattur. It’s been a week since my hair procedure with you in Mumbai. The recovery has been going well and I am back to the grind of my every day routine. I want to take this opportunity to thank you and your dear wife who went the extra mile to accommodate me given my short read more...
I first contacted Dr. Sandeep Sattur and Dr. Indu Sattur back in 2013, when I was keen on finding out what’s involved in a hair transplant procedure. On top of my head, hairs was receding at a steady rate. Dr. Sandeep Sattur and Dr. Indu Sattur, cleared all of my doubts and after read more...
The whole trip was so quick that it seems like a dream. It is comforting that I was in the professional care of you and your friendly staff.–Everyone who visits your website should be aware of this! If your objective is to attract a foreign patient base- I think the first thing read more...
Our Team

DR. SANDEEP SATTUR
Founder, Hair Transplant Surgeon, Plastic Surgeon.
Hairrevive - Centre for hair restoration and skin rejuvenation
An alumnus of the Grant Medical College, Mumbai, India. He completed his MBBS, MS (General Surgery) and M.Ch. (Plastic Surgery) from this institute.
He has been practicing in Mumbai for the last 18 years being affiliated to and having hospital privileges at Dr. L H Hiranandani Hospital Powai, and Wockhardt Hospital, Mumbai Central.

DR. INDU SATTUR
Medical Director, Hair Restoration Specialist and Cosmetologist at Hairrevive – Center for Hair Restoration and Skin Rejuvenation.
An alumnus of Grant Medical College, Mumbai, India, she completed her MBBS and Diploma in Anaesthesia from this institute. Initial years were dedicated to the practice of anesthesia. However, she was a part of the hair transplant team right from the beginning. Due to her interest in Cosmetic Science and Hair Restoration, she choose to opt for a practice dedicated to cosmetology and hair restoration. Moving ahead in this direction she completed her Diploma in Trichology and Skin Aesthetics.

DR. SURUCHI PATIL
Associate, Hair Restoration, and Cosmetology at Hairrevive – Center for Hair Restoration and Skin Rejuvenation.
Dr. Suruchi Patil is an alumnus of Nagpur University, who is a part of the Hair Transplant team at Hairrevive. She assists in hair transplant. She also is involved in performing Chemical peels, Microdermabrasion, and LHR.